Survivor Views: Telehealth and Clinical Trials
Overview:
The American Cancer Society Cancer Action Network (ACS CAN) gives voice to cancer patients and survivors on critical public policy issues that affect their lives. As part of this effort, ACS CAN deploys surveys to better understand cancer patient and survivor experiences and perspectives, through our Survivor Views research panel. The panel is a group of cancer patients and survivors who respond to regular surveys and provide important insights to support ACS CAN’s public policy work at all levels of government.
Fielded July 6 to September 8, 2021, the latest survey explores telehealth experiences and preferences, along with interest and willingness to participate in clinical trials. The web-based survey was completed by 1,344 patients and survivors diagnosed with or treated for cancer in the last seven years. The results provide important insights into the experiences and opinions of cancer patients and survivors in accessing care remotely, which has the potential to improve alternate routes to access care and participate in clinical trials.
Key Findings:
- Overwhelming majorities of cancer patients and survivors who have had telehealth visits believed their issues and questions were well-addressed. Fifty-five percent of respondents have had a phone visit and 43 percent have had a video visit with a telehealth provider about an issue related to their cancer care that otherwise would have been an in-person office visit (not a prescription refill or appointment booking). In both cases, 94% said their issues and questions were addressed well.
- Cancer survivors and patients are very receptive to various technologies and methods that facilitate remote care. More than eighty percent of respondents would be willing to adopt several telehealth technologies, including using health apps to monitor personal health data, sharing data collected from health apps, using wearable technology to monitor personal health data, and sharing the data collected by wearables with their providers.
- Although a plurality have never discussed trial participation with their provider, most would like to join a trial if participation was at least as easy as their regular care. Nearly half of respondents would like to participate even if it required extra effort in terms of more frequent visits or additional travel. Higher income earners are significantly more likely to say they would want to participate even if it required extra effort.
- Trial sponsor support to offset travel costs increases willingness to enroll in clinical trials. Over three-quarters of cancer patients and survivors would be more likely to participate in a clinical trial located outside of their local area if the trial sponsor helped to organize or pay for travel and lodging to offset the extra travel.
Detailed Survey Findings:
Patient Experiences with Telehealth are Overwhelmingly Positive
At this point in the pandemic, many cancer patients and survivors have already had telehealth visits. Over half (55%) have had a phone visit and 43% have had a video visit with a telehealth provider about an issue related to their cancer care that otherwise would have required an in-person office visit (i.e. not a prescription refill or appointment booking). While those who receive care in an urban setting are more likely to have had a video visit with a provider (55%), those treated in rural settings have made up the difference with more phone visits (64%). In both cases, 94% said their issues and questions were addressed well, with two-thirds saying they were addressed very well.
Figure 1: Cancer Patients’ Needs Well Met by Televisits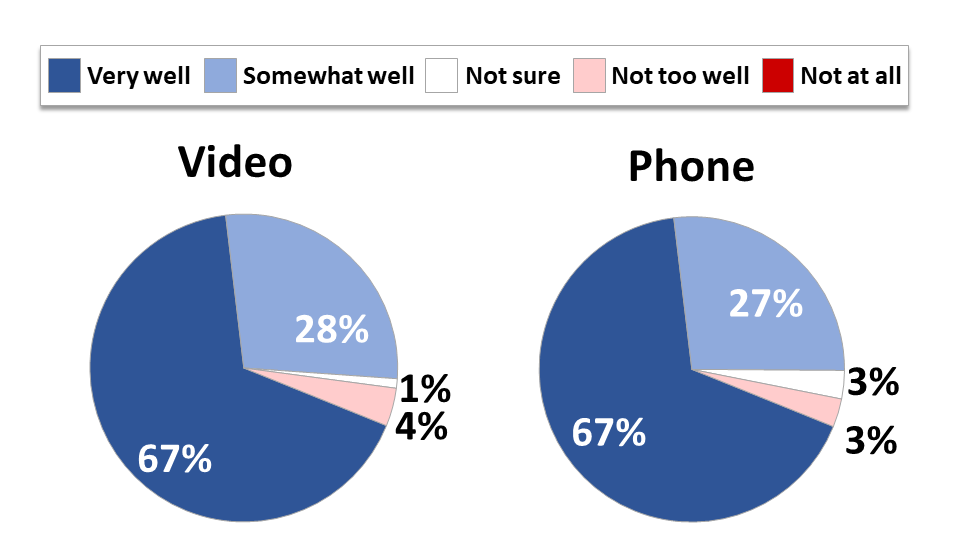
These current numbers reflect a significant increase in telehealth use compared to before the pandemic. Just 38% of respondents to the October 2019 Survivor Views survey reported having a phone visit with a provider, and only 1% had had a video visit. Although many have now had telehealth experiences, 42% of those who have not yet had a video visit with a provider said they would like to, bringing the total who have had or would like to have a video visit to two-thirds. While the majority of respondents who had a video visit were able to do so without technical assistance, one-quarter required some assistance setting up or interacting with the equipment. Those who needed technical assistance were more likely to be lower income, less educated, younger, racial and ethnic minorities, and female, suggesting that offering adequate technical support is important to reducing telehealth access barriers that disproportionately impact certain demographics.
Strong Willingness to Adopt Technologies and Methods that Facilitate Remote Care
More than 80% of respondents would be willing to adopt several telehealth technologies if they were provided to them. The use of health apps to monitor personal health data (84%), sharing data collected from these health apps with providers (84%), using wearable technology to monitor personal health data (84%), and sharing the data collected by wearables with their providers (83%) were all well-received, with majorities saying they would be very willing to adopt these technologies if they were made available to them. Patients’ strong willingness to provide informed consent electronically (88%) further demonstrates their comfort with digital accommodations to remote care.
Figure 2: Strong Willingness to Adopt Telehealth Accommodations
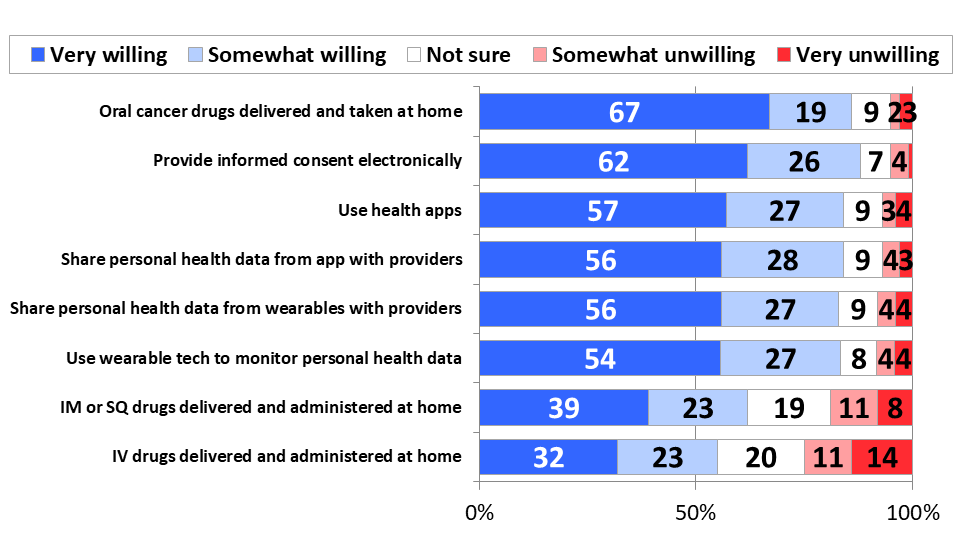
The most popular remote care adaptation, however, removes the inconvenience of pharmacy visits. Eighty-eight percent of patients are eager to have oral cancer drugs delivered to them and taken at home, with 67% very willing and only 5% unwilling. Majorities also would be willing to receive intramuscular and subcutaneous injections (62%), and even IV drugs at home (55%), but by decreasing margins.
Most Have Never Discussed Trial Participation, but Majority Would Want to Join One
Less than one-in-five of those surveyed have participated in a cancer clinical trial (18%), and nearly half (46%) have never discussed it with their provider. Another 17% discussed trials generally with their provider, but were never offered the opportunity to participate. Just 7% declined to participate in a trial they qualified for, while a similar number looked for a trial on their own but did not ultimately enroll in one (6%).
Figure 3: Trial Experience
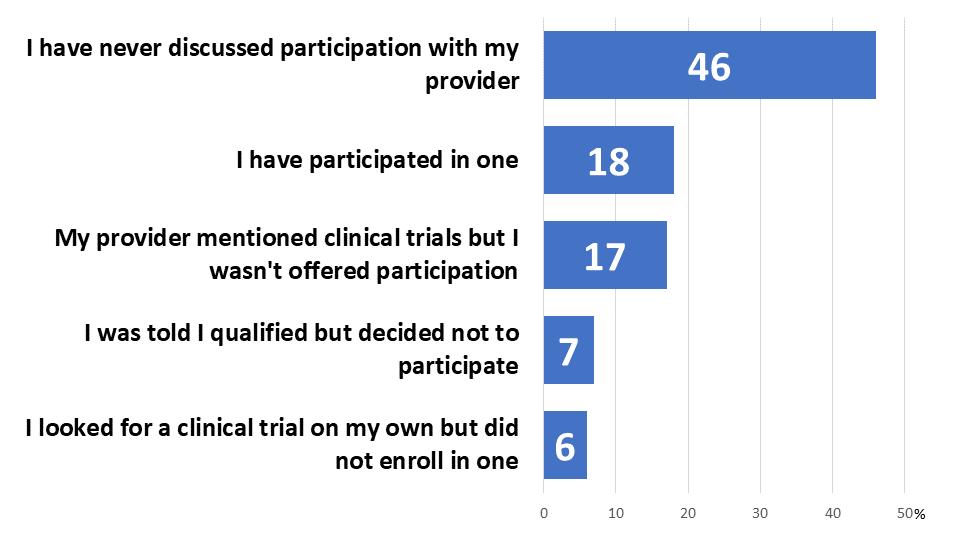
While a plurality have never discussed participation with a provider, most would like to join a cancer clinical trial if it was at least as easy as their regular care (77%). Furthermore, nearly half would like to participate even if it required extra effort in terms of more frequent visits or additional travel (47%). Only 7% would hold out for a trial that was easier than their usual care, while 14% would need an “extremely compelling reason” and 2% would not join a cancer clinical trial under any circumstance. Higher income earners are significantly more likely to say they would want to participate in a trial that required extra effort (62% of those earning over $125,000 annually compared to 41% of those earning $70,000 or less). Since many trials do require extra effort, this suggests practical barriers that are keeping otherwise willing, low-income patients from participating.
Figure 4: Trial Willingness
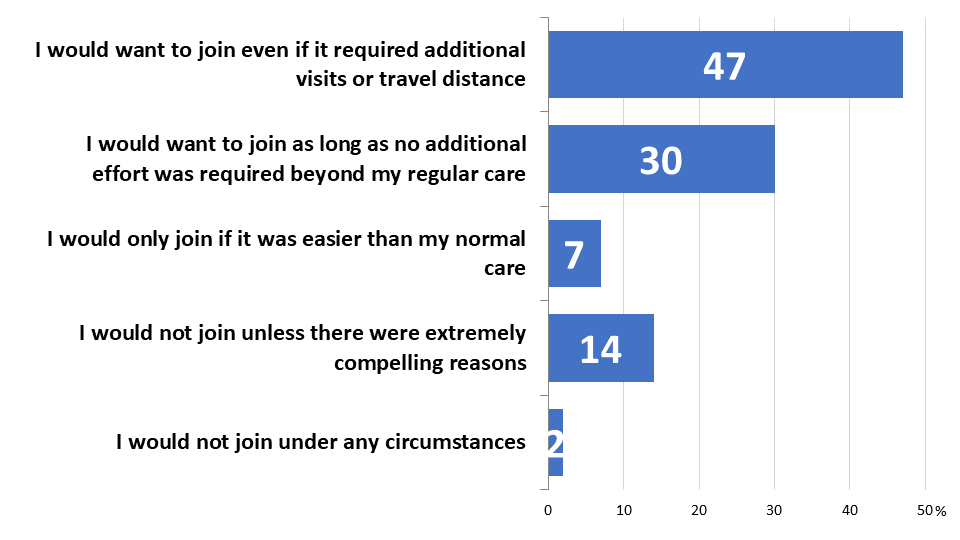
Majorities Are Willing to Travel in Order to Participate in Trials, But Not All Are Equally Able
Further demonstrating cancer survivors’ and patients’ eagerness to participate in cancer clinical trials, over three-quarters would be willing to drive 30 minutes or more further than their regular care in order to participate (77%), and 30% would travel an additional 90 minutes or more. Twenty percent would be willing to participate in a trial that required overnight travel. For context, 32% of respondents currently travel within ten miles of their home for their regular cancer care, and 68% travel within 24 miles of home. Fewer than one-in-five travel more than an hour to reach their regular cancer care provider (18%).
Figure 5: Willingness to Travel to Participate in Trials
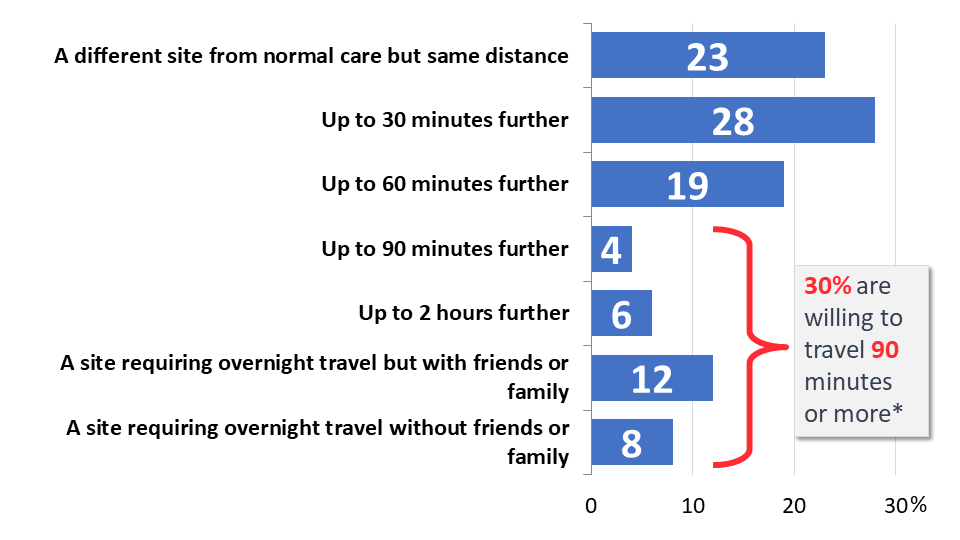
However, willingness to travel greater distances decreases with reduced income, creating potential disparities in clinical trial participation. Thirty-six percent of those earning over $125,000 annually would be willing to travel 90 minutes or more further than their regular care, compared to 27% of those earning less than $70,000 per year.
Trial Sponsor Support to Offset Extra Travel is Important Across Demographic Groups
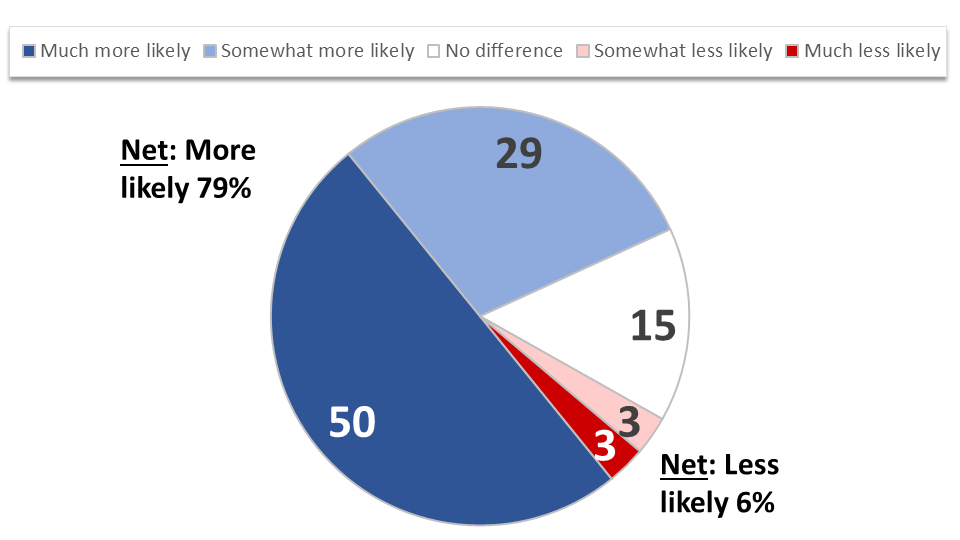
Additionally, 79% of cancer patients and survivors say they would be more likely to participate in a clinical trial located outside of their local area if the trial sponsor helped to organize or pay for travel and lodging, and 50% would be much more likely. This finding is consistent across many demographic variables including income and whether the respondent lives or receives care in an urban, suburban, or rural setting. While higher income earners are slightly more likely to say sponsor support of trial-related travel and lodging would not make a difference in their willingness to participate (17%), offsetting these costs is important across all income groups. Those in their prime earning years (ages 35 to 54) are most likely to report increased likelihood of participation with such support (63% much more likely).
Figure 6: Trial Sponsor Support Increases Likelihood of Participation
Methodology:
ACS CAN’s Survivor Views research initiative was designed to enhance the organization’s mission to end suffering and death from cancer. Data provided by cancer patients and survivors as part of this project allows for a greater understanding of their experiences and opinions on cancer-related issues and gives voice to cancer patients and survivors in the shaping and advocating of public policies that help prevent, detect, and treat cancer and promote a more positive quality of life for those impacted.
To ensure the protection of all participants in this initiative all research protocols, questionnaires, and communications are reviewed by the Morehouse School of Medicine Institutional Review Board.
The survey population is comprised of individuals who meet the following criteria:
- Diagnosed with and/or treated for cancer within the last seven years
- Over the age of 18 (parents of childhood cancer survivors were invited to participate on behalf of their minor children)
- Reside in the US or US territories
Potential Survivor Views participants were invited to participate through email invitations, social media promotion, and partner group outreach. Those who agreed to participate after reviewing the informed consent information completed a brief survey including demographic and cancer history information to inform analysis as well as topical questions as discussed in this document. The data were collected between July 6 and September 8, 2021. A total of 1,344 participants responded to the survey, providing a margin of error +/- 2.7% at a 96% confidence level.
About ACS CAN
The American Cancer Society Cancer Action Network (ACS CAN) is making cancer a top priority for public officials and candidates at the federal, state and local levels. ACS CAN empowers advocates across the country to make their voices heard and influence evidence-based public policy change as well as legislative and regulatory solutions that will reduce the cancer burden. As the American Cancer Society’s nonprofit, nonpartisan advocacy affiliate, ACS CAN is critical to the fight for a world without cancer. For more information, visit www.fightcancer.org.